200,000+ products from a single source!
sales@angenechem.com
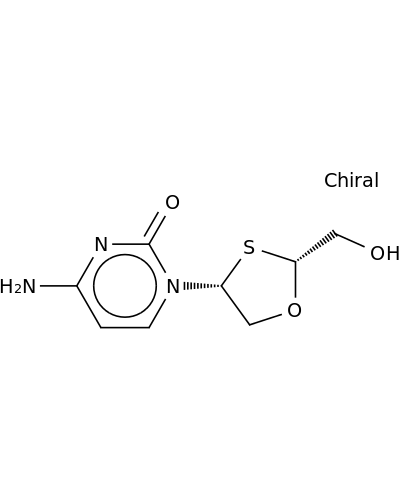
160707-69-7 | 2(1H)-Pyrimidinone, 4-amino-1-[(2R,4R)-2-(hydroxymethyl)-1,3-oxathiolan-4-yl]-
CAS No: 160707-69-7 Catalog No: AG001S4O MDL No:
Product Description
Catalog Number:
AG001S4O
Chemical Name:
2(1H)-Pyrimidinone, 4-amino-1-[(2R,4R)-2-(hydroxymethyl)-1,3-oxathiolan-4-yl]-
CAS Number:
160707-69-7
Molecular Formula:
C8H11N3O3S
Molecular Weight:
229.2562
IUPAC Name:
4-amino-1-[(2R,4R)-2-(hydroxymethyl)-1,3-oxathiolan-4-yl]pyrimidin-2-one
InChI:
InChI=1S/C8H11N3O3S/c9-5-1-2-11(8(13)10-5)6-4-14-7(3-12)15-6/h1-2,6-7,12H,3-4H2,(H2,9,10,13)/t6-,7-/m1/s1
InChI Key:
RYMCFYKJDVMSIR-RNFRBKRXSA-N
SMILES:
Nc1ccn(c(=O)n1)[C@H]1CO[C@H](S1)CO
UNII:
K1YX059ML1
Properties
Complexity:
331
Compound Is Canonicalized:
Yes
Covalently-Bonded Unit Count:
1
Defined Atom Stereocenter Count:
2
Defined Bond Stereocenter Count:
0
Exact Mass:
229.052g/mol
Formal Charge:
0
Heavy Atom Count:
15
Hydrogen Bond Acceptor Count:
4
Hydrogen Bond Donor Count:
2
Isotope Atom Count:
0
Molecular Weight:
229.254g/mol
Monoisotopic Mass:
229.052g/mol
Rotatable Bond Count:
2
Topological Polar Surface Area:
113A^2
Undefined Atom Stereocenter Count:
0
Undefined Bond Stereocenter Count:
0
XLogP3:
-1.2
Literature
| Title | Journal |
|---|---|
| Antiretroviral drugs: critical issues and recent advances. | Indian journal of pharmacology 20120101 |
| Renal excretion of apricitabine in rats: ex vivo and in vivo studies. | European journal of drug metabolism and pharmacokinetics 20110901 |
| Antiviral activity of apricitabine in treatment-experienced HIV-1-infected patients with M184V who are failing combination therapy. | HIV medicine 20110701 |
| Recent advances in antiretroviral drugs. | Expert opinion on pharmacotherapy 20110101 |
| A template-dependent dislocation mechanism potentiates K65R reverse transcriptase mutation development in subtype C variants of HIV-1. | PloS one 20110101 |
| Resistance profile of the new nucleoside reverse transcriptase inhibitor apricitabine. | The Journal of antimicrobial chemotherapy 20100201 |
| Apricitabine: a nucleoside reverse transcriptase inhibitor for HIV infection. | The Annals of pharmacotherapy 20091001 |
| Gateways to clinical trials. | Methods and findings in experimental and clinical pharmacology 20090401 |
| Apricitabine--a novel nucleoside reverse transcriptase inhibitor for the treatment of HIV infection that is refractory to existing drugs. | Expert opinion on investigational drugs 20090201 |
| An analysis of enzyme kinetics data for mitochondrial DNA strand termination by nucleoside reverse transcription inhibitors. | PLoS computational biology 20090101 |
| Effects of the K65R and K65R/M184V reverse transcriptase mutations in subtype C HIV on enzyme function and drug resistance. | Retrovirology 20090101 |
| Novel compounds for the treatment of HIV type-1 infection. | Antiviral chemistry & chemotherapy 20090101 |
| Pharmacotherapy of pediatric and adolescent HIV infection. | Therapeutics and clinical risk management 20090101 |
| Comparison of the pharmacokinetics of apricitabine in the presence and absence of ritonavir-boosted tipranavir: a phase I, open-label, controlled, single-centre study. | Clinical drug investigation 20090101 |
| Emerging antiviral drugs. | Expert opinion on emerging drugs 20080901 |
| Apricitabine continues to show good results. | Project Inform perspective 20080901 |
| Influence of food on the pharmacokinetics of apricitabine, a novel deoxycytidine analogue reverse transcriptase inhibitor. | Expert opinion on pharmacotherapy 20080801 |
| New drugs. | Journal of HIV therapy 20080601 |
| Pharmacokinetics of apricitabine, a novel nucleoside reverse transcriptase inhibitor, in healthy volunteers treated with trimethoprim-sulphamethoxazole. | Journal of clinical pharmacy and therapeutics 20080201 |
| Multiple-dose pharmacokinetics of apricitabine, a novel nucleoside reverse transcriptase inhibitor, in patients with HIV-1 infection. | Clinical drug investigation 20080101 |
| Effects of apricitabine and other nucleoside reverse transcriptase inhibitors on replication of mitochondrial DNA in HepG2 cells. | Antiviral research 20071001 |
| Gateways to clinical trials. | Methods and findings in experimental and clinical pharmacology 20070101 |
| Apricitabine: a novel deoxycytidine analogue nucleoside reverse transcriptase inhibitor for the treatment of nucleoside-resistant HIV infection. | Antiviral chemistry & chemotherapy 20070101 |
| Kinetics of inhibition of HIV type 1 reverse transcriptase-bearing NRTI-associated mutations by apricitabine triphosphate. | Antiviral chemistry & chemotherapy 20070101 |
| Effects of trimethoprim on the clearance of apricitabine, a deoxycytidine analog reverse transcriptase inhibitor, and Lamivudine in the isolated perfused rat kidney. | The Journal of pharmacology and experimental therapeutics 20061101 |
| Efficacy and tolerability of 10-day monotherapy with apricitabine in antiretroviral-naive, HIV-infected patients. | AIDS (London, England) 20060612 |
| In vitro antiretroviral activity and in vitro toxicity profile of SPD754, a new deoxycytidine nucleoside reverse transcriptase inhibitor for treatment of human immunodeficiency virus infection. | Antimicrobial agents and chemotherapy 20060201 |
| Pharmacokinetics of single oral doses of apricitabine, a novel deoxycytidine analogue reverse transcriptase inhibitor, in healthy volunteers. | Clinical drug investigation 20060101 |
| New drugs. | HIV medicine 20050701 |
| In vitro activity of structurally diverse nucleoside analogs against human immunodeficiency virus type 1 with the K65R mutation in reverse transcriptase. | Antimicrobial agents and chemotherapy 20050301 |
| In vitro activity of SPD754, a new deoxycytidine nucleoside reverse transcriptase inhibitor (NRTI), against 215 HIV-1 isolates resistant to other NRTIs. | Antiviral chemistry & chemotherapy 20050101 |
| Selection of resistance-conferring mutations in HIV-1 by the nucleoside reverse transcriptase inhibitors (+/-)dOTC and (+/-)dOTFC. | Antiviral chemistry & chemotherapy 20001101 |
| Drug resistance and drug combination features of the human immunodeficiency virus inhibitor, BCH-10652 [(+/-)-2'-deoxy-3'-oxa-4'-thiocytidine, dOTC]. | Antiviral chemistry & chemotherapy 20000701 |
| Selection and characterization of human immunodeficiency virus type 1 variants resistant to the (+) and (-) enantiomers of 2'-deoxy-3'-oxa-4'-thio-5-fluorocytidine. | Antimicrobial agents and chemotherapy 20000501 |
| Antiviral activity of 2'-deoxy-3'-oxa-4'-thiocytidine (BCH-10652) against lamivudine-resistant human immunodeficiency virus type 1 in SCID-hu Thy/Liv mice. | Antimicrobial agents and chemotherapy 20000301 |
| Anti-human immunodeficiency virus and anti-hepatitis-B virus activities and toxicities of the enantiomers of 2'-deoxy-3'-oxa-4'-thiocytidine and their 5-fluoro analogues in vitro. | Journal of medicinal chemistry 19950106 |
Related Products
© 2019 Angene International Limited. All rights Reserved.