200,000+ products from a single source!
sales@angenechem.com
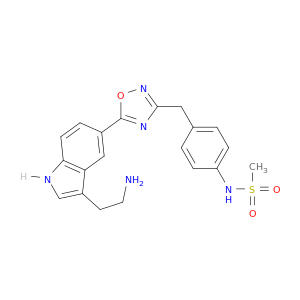
137403-12-4 | Methanesulfonamide, N-[4-[[5-[3-(2-aminoethyl)-1H-indol-5-yl]-1,2,4-oxadiazol-3-yl]methyl]phenyl]-
CAS No: 137403-12-4 Catalog No: AG00138N MDL No:MFCD00924342
Product Description
Catalog Number:
AG00138N
Chemical Name:
Methanesulfonamide, N-[4-[[5-[3-(2-aminoethyl)-1H-indol-5-yl]-1,2,4-oxadiazol-3-yl]methyl]phenyl]-
CAS Number:
137403-12-4
Molecular Formula:
C20H21N5O3S
Molecular Weight:
411.4774
MDL Number:
MFCD00924342
IUPAC Name:
N-[4-[[5-[3-(2-aminoethyl)-1H-indol-5-yl]-1,2,4-oxadiazol-3-yl]methyl]phenyl]methanesulfonamide
InChI:
InChI=1S/C20H21N5O3S/c1-29(26,27)25-16-5-2-13(3-6-16)10-19-23-20(28-24-19)14-4-7-18-17(11-14)15(8-9-21)12-22-18/h2-7,11-12,22,25H,8-10,21H2,1H3
InChI Key:
HKXMQLISPYELRD-UHFFFAOYSA-N
SMILES:
NCCc1c[nH]c2c1cc(cc2)c1onc(n1)Cc1ccc(cc1)NS(=O)(=O)C
Properties
Complexity:
635
Compound Is Canonicalized:
Yes
Covalently-Bonded Unit Count:
1
Defined Atom Stereocenter Count:
0
Defined Bond Stereocenter Count:
0
Exact Mass:
411.137g/mol
Formal Charge:
0
Heavy Atom Count:
29
Hydrogen Bond Acceptor Count:
7
Hydrogen Bond Donor Count:
3
Isotope Atom Count:
0
Molecular Weight:
411.48g/mol
Monoisotopic Mass:
411.137g/mol
Rotatable Bond Count:
7
Topological Polar Surface Area:
135A^2
Undefined Atom Stereocenter Count:
0
Undefined Bond Stereocenter Count:
0
XLogP3:
2.2
Literature
| Title | Journal |
|---|---|
| Serotonin modulates glutamatergic transmission in the rat olfactory tubercle. | The European journal of neuroscience 20100201 |
| Peripheral 5-HT1A and 5-HT7 serotonergic receptors modulate parasympathetic neurotransmission in long-term diabetic rats. | Experimental diabetes research 20100101 |
| Sumatriptan inhibits synaptic transmission in the rat midbrain periaqueductal grey. | Molecular pain 20080101 |
| Diabetes-induced changes in 5-hydroxytryptamine modulation of vagally-induced bradycardia in rat heart. | Clinical and experimental pharmacology & physiology 20071101 |
| The nitric oxide synthesis/pathway mediates the inhibitory serotoninergic responses of the pressor effect elicited by sympathetic stimulation in diabetic pithed rats. | European journal of pharmacology 20060510 |
| 5-HT-stimulated [35S]guanosine-5'-O-(3-thio)triphosphate binding as an assay for functional activation of G proteins coupled with 5-HT1B receptors in rat striatal membranes. | Naunyn-Schmiedeberg's archives of pharmacology 20060201 |
| Effect of 5-hydroxytryptamine on neurogenic vasoconstriction in the isolated, autoperfused hindquarters of the rat. | Clinical and experimental pharmacology & physiology 20051001 |
| In vivo effects of local activation and blockade of 5-HT1B receptors on globus pallidus neuronal spiking. | Brain research 20050510 |
| Specific labelling of serotonin 5-HT(1B) receptors in rat frontal cortex with the novel, phenylpiperazine derivative, [3H]GR125,743. A pharmacological characterization. | Pharmacology, biochemistry, and behavior 20020401 |
| Molecular cloning and expression of the porcine trigeminal ganglion cDNA encoding a 5-ht(1F) receptor. | European journal of pharmacology 20020201 |
| The proliferation of human T lymphoblastic cells induced by 5-HT1B receptors activation is regulated by 5-HT-moduline. | Comptes rendus de l'Academie des sciences. Serie III, Sciences de la vie 20010401 |
| Identification of an amino acid residue important for binding of methiothepin and sumatriptan to the human 5-HT(1B) receptor. | European journal of pharmacology 19990910 |
| Chimeric receptor analysis of the ketanserin binding site in the human 5-Hydroxytryptamine1D receptor: importance of the second extracellular loop and fifth transmembrane domain in antagonist binding. | Molecular pharmacology 19981201 |
| Agonist activity of antimigraine drugs at recombinant human 5-HT1A receptors: potential implications for prophylactic and acute therapy. | Naunyn-Schmiedeberg's archives of pharmacology 19970601 |
Related Products
© 2019 Angene International Limited. All rights Reserved.