200,000+ products from a single source!
sales@angenechem.com
Home > Thiazole > 1004316-88-4
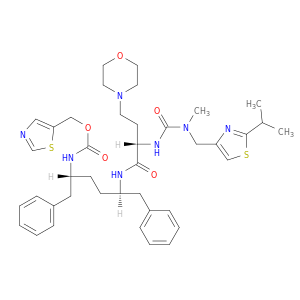
1004316-88-4 | 2,7,10,12-Tetraazatridecanoic acid, 12-methyl-13-[2-(1-methylethyl)-4-thiazolyl]-9-[2-(4-morpholinyl)ethyl]-8,11-dioxo-3,6-bis(phenylmethyl)-, 5-thiazolylmethyl ester, (3R,6R,9S)-
CAS No: 1004316-88-4 Catalog No: AG0001ZB MDL No:MFCD18251449
Product Description
Catalog Number:
AG0001ZB
Chemical Name:
2,7,10,12-Tetraazatridecanoic acid, 12-methyl-13-[2-(1-methylethyl)-4-thiazolyl]-9-[2-(4-morpholinyl)ethyl]-8,11-dioxo-3,6-bis(phenylmethyl)-, 5-thiazolylmethyl ester, (3R,6R,9S)-
CAS Number:
1004316-88-4
Molecular Formula:
C40H53N7O5S2
Molecular Weight:
776.0227
MDL Number:
MFCD18251449
IUPAC Name:
1,3-thiazol-5-ylmethyl N-[(2R,5R)-5-[[(2S)-2-[[methyl-[(2-propan-2-yl-1,3-thiazol-4-yl)methyl]carbamoyl]amino]-4-morpholin-4-ylbutanoyl]amino]-1,6-diphenylhexan-2-yl]carbamate
InChI:
InChI=1S/C40H53N7O5S2/c1-29(2)38-43-34(27-53-38)25-46(3)39(49)45-36(16-17-47-18-20-51-21-19-47)37(48)42-32(22-30-10-6-4-7-11-30)14-15-33(23-31-12-8-5-9-13-31)44-40(50)52-26-35-24-41-28-54-35/h4-13,24,27-29,32-33,36H,14-23,25-26H2,1-3H3,(H,42,48)(H,44,50)(H,45,49)/t32-,33-,36+/m1/s1
InChI Key:
ZCIGNRJZKPOIKD-CQXVEOKZSA-N
SMILES:
O=C(N[C@@H](Cc1ccccc1)CC[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)N(Cc1csc(n1)C(C)C)C)CCN1CCOCC1)OCc1cncs1
UNII:
LW2E03M5PG
Properties
Complexity:
1120
Compound Is Canonicalized:
Yes
Covalently-Bonded Unit Count:
1
Defined Atom Stereocenter Count:
3
Defined Bond Stereocenter Count:
0
Exact Mass:
775.355g/mol
Formal Charge:
0
Heavy Atom Count:
54
Hydrogen Bond Acceptor Count:
10
Hydrogen Bond Donor Count:
3
Isotope Atom Count:
0
Molecular Weight:
776.028g/mol
Monoisotopic Mass:
775.355g/mol
Rotatable Bond Count:
20
Topological Polar Surface Area:
195A^2
Undefined Atom Stereocenter Count:
0
Undefined Bond Stereocenter Count:
0
XLogP3:
5.7
Literature
| Title | Journal |
|---|---|
| Pharmacokinetic enhancers in HIV therapeutics. | Clinical pharmacokinetics 20141001 |
| Cobicistat boosts the intestinal absorption of transport substrates, including HIV protease inhibitors and GS-7340, in vitro. | Antimicrobial agents and chemotherapy 20121001 |
| Effect of cobicistat on glomerular filtration rate in subjects with normal and impaired renal function. | Journal of acquired immune deficiency syndromes (1999) 20120901 |
| Combinational therapies for HIV: a focus on EVG/COBI/FTC/TDF. | Expert opinion on pharmacotherapy 20120901 |
| [A single tablet against HIV: new combination preparation improves therapy]. | Deutsche medizinische Wochenschrift (1946) 20120801 |
| Co-formulated elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate versus ritonavir-boosted atazanavir plus co-formulated emtricitabine and tenofovir disoproxil fumarate for initial treatment of HIV-1 infection: a randomised, double-blind, phase 3, non-inferiority trial. | Lancet (London, England) 20120630 |
| Co-formulated elvitegravir, cobicistat, emtricitabine, and tenofovir versus co-formulated efavirenz, emtricitabine, and tenofovir for initial treatment of HIV-1 infection: a randomised, double-blind, phase 3 trial, analysis of results after 48 weeks. | Lancet (London, England) 20120630 |
| Phase 2 study of cobicistat versus ritonavir each with once-daily atazanavir and fixed-dose emtricitabine/tenofovir df in the initial treatment of HIV infection. | AIDS (London, England) 20110924 |
| Randomized, phase 2 evaluation of two single-tablet regimens elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil fumarate versus efavirenz/emtricitabine/tenofovir disoproxil fumarate for the initial treatment of HIV infection. | AIDS (London, England) 20110327 |
| Pharmacokinetics and bioavailability of an integrase and novel pharmacoenhancer-containing single-tablet fixed-dose combination regimen for the treatment of HIV. | Journal of acquired immune deficiency syndromes (1999) 20101101 |
| Cobicistat (GS-9350): A Potent and Selective Inhibitor of Human CYP3A as a Novel Pharmacoenhancer. | ACS medicinal chemistry letters 20100812 |
| Pharmacokinetics and pharmacodynamics of GS-9350: a novel pharmacokinetic enhancer without anti-HIV activity. | Clinical pharmacology and therapeutics 20100301 |
Related Products
© 2019 Angene International Limited. All rights Reserved.